Abstract
Background
Data from randomized-controlled trials (RCT) do not necessarily represent expected outcomes in real-world experience due to constraints of strict enrollment criteria and protocol-directed allowable treatments. Real-world data (RWD), while less well-controlled and thus less specific to a given patient population, incorporate clinical experience, patient preferences, and real-world issues that are a routine part of clinical practice and thus pertinent to patient care. Thus, RWD complement results obtained in an RCT. Fostamatinib is an orally administered spleen tyrosine kinase (SYK) inhibitor indicated for treatment adults with chronic ITP who had an insufficient response to previous therapy. The objective of this analysis was to evaluate real-world patient-reported outcomes from patients with ITP treated with fostamatinib and enrolled in a patient support program (PSP).
Methods
Patients were included if they had: an ICD10 code consistent with ITP diagnosis; ≥1 pre-treatment platelet counts reported by the provider +/- patient; ≥1 post-fostamatinib initiation platelet counts reported by the patient; ≥3 months of drug supply with ≤60-day gap during the treatment period; and signed a written consent to share data. Patients were registered by their physician for the manufacturer-sponsored PSP. The PSP registration document includes a form to collect baseline demographic and ITP-specific data. A PSP nurse navigator then calls patients every two weeks (or as agreed upon) to provide support and resources to patients regarding insurance coverage, medication adherence, and how/when to contact their provider. Information on affordability and access to programs such as copay/coinsurance assistance and free drug supply in cases where patients meet the program criteria are also discussed. During the calls, the nurse navigator collects any platelet counts reported by the patient.
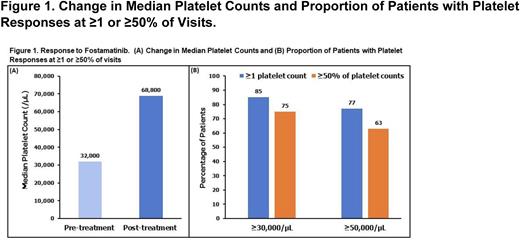
Pre-specified study endpoints include median post-treatment platelet counts and the proportion of patients who achieved 1) platelet counts of ≥30,000/µL or ≥50,000/µL, 2) platelet counts of ≥30,000/µL or ≥50,000/µL on ≥50% of post-treatment readings, and/or 3) ≥1 post-treatment doubling of platelet count from the pre-treatment median.
Results
As of the data cutoff date (10 March 2022), 318 patients were included: 108 (34%) patients remained on 100 mg BID, and 210 patients (66%) had increased their dose to 150 mg BID at some point during treatment. The median age was 67 (range 19-100), and mean duration of fostamatinib therapy was 11 months. The median pre-treatment platelet counts (32,000/µL) were higher than those in the RCT (16,000/µL), possibly due to patients receiving rescue treatment or switching to fostamatinib from other maintenance therapies without the washout period required in the clinical trials. Most patients (85%) achieved platelet counts of ≥30,000/µL at least once (compared with 70% in the RCT), and 75% had at least half their counts ≥30,000/µL. Interestingly, this RWD also demonstrated a higher proportion of patients (77%) with counts ≥50,000/µL (compared with 54% in the RCT), and 63% had at least half their counts ≥50,000/µL (Figure 1). Most patients (65%) demonstrated a doubling of their median pre-treatment platelet count.
Limitations include that this report did not evaluate safety. Additionally, the potential for reporter bias exists since patients self-reported post-treatment platelet counts. Pre-treatment platelet counts were reported by the healthcare provider on the enrollment form and may have been supplemented by patient-reported data.
Conclusions
In this analysis of RWD from patients supported by Rigel OneCare®, fostamatinib demonstrated substantial efficacy in clinical practice. These results complement and amplify those of the clinical trial program. They further support the use of fostamatinib for management of ITP.
Disclosures
Bussel:AstraZeneca: Other: Data and Safety Monitoring Board; Rallybio: Consultancy, Membership on an entity's Board of Directors or advisory committees; Rigel: Consultancy, Membership on an entity's Board of Directors or advisory committees; Argenx: Consultancy, Membership on an entity's Board of Directors or advisory committees; Amgen: Consultancy, Membership on an entity's Board of Directors or advisory committees; Sobi: Consultancy, Membership on an entity's Board of Directors or advisory committees; UCB: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: Data and Safety Monitoring Board; Novartis: Consultancy, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Janssen: Consultancy, Membership on an entity's Board of Directors or advisory committees. Hales:Rigel Pharmaceuticals, Inc.: Current Employment, Current equity holder in publicly-traded company. Buxman:Claritas, LLC.: Current Employment. Todd:Rigel Pharmaceuticals, Inc.: Current Employment, Current equity holder in publicly-traded company.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal